Cardio Diagnostics (CDIO) Updates
The Long-term Value Thesis Remains Intact
The purpose of this update on CDIO is mainly to focus on the Feb 18th investor conference call. There have been some key recent developments too. Here is my original write-up on the company last summer.
In January Cardio Diagnostics announced the Centers for Medicare and Medicaid Services (CMS) is paying $854 for both the Epi+Gen CHD™ and PrecisionCHD™ tests. This is more than last years preliminary estimates of $350 for Epi+Gen CHD™ and $684.76 for PrecisionCHD™.
The company partnered with Dr. Lal PathLabs in India who have 290 clinical laboratories and 300 MD pathologists who will be using the PrecisionCHD™ tests.
There is new data from a study titled "DNA Methylation Strongly Predicts Level of Coronary Artery Obstruction and Ischemia with No Obstructive Coronary Artery Spectrum Presentations in Those with Acute Coronary Syndrome."
The findings build on the previous data in the Journal of the American Heart Association validating PrecisionCHD’s ability to sensitively and specifically detect coronary heart disease.
Highlights From The February Investor Call
In the beginning of the call, the chairman essentially proposed the question people ask when they discover the company. If there is real value in the test where is the revenue? He goes on to talk about how it’s a long process and uses Cologuard an epigenetic test, that was launched in 2014 and wasn’t bought by Abbott Labs until 2025. I see Cardio Diagnostics value being realized much faster due to the advances in AI the last few years and the superior nature of AI combined with medicine.
The PrecisionCHD test shows higher sensitivity and specificity at detecting coroanary heart disease than an exercise stress treadmill.
They have developed the first and only commercial solutions using epigenetics and AI for cardiovascular disease prevention and early detection.
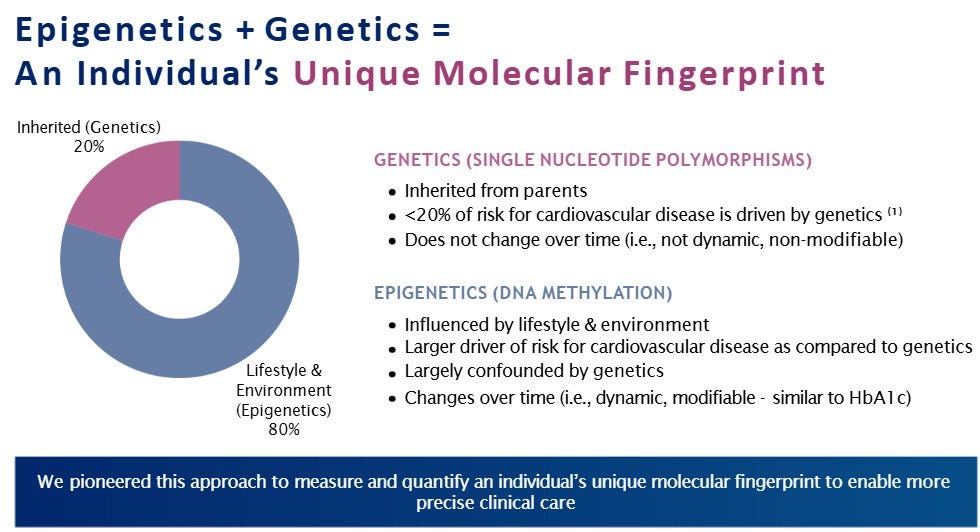
Surprising to me the CEO mentioned genetics is only 20% of cardiovascular risk with it mainly being epigenetic. See below
This role of genetics is the consensus among major health organizations like the CDC and WHO.
They have a proprietary AI algorithm that identifies and integrates each patients unique biomarkers. The lab testing was built over 14 years and can be expanded upon.
The CEO says their tests have a cost savings of $42,000 per quality adjusted life year. 146 million Americans are elligible for their EPI+ test. 60 million Americans are eligible for the Precision CHD test. They estimate $113 million in cost savings to insurers and employers in the first year for the Precision CHD test.
It’s not a one and done test. The tests can be repeated over time.
The CEO says they are seeing growth in their sales pipeline and interest in the tests.
With the stock being up around 300%+ the past month I expect an offering.
I continue to like CDIO based on the thematic and possibly large upside over time or a buyout. I went into some speculation on a buyout in the write-up linked up top.
Full Disclosure: I am long CDIO shares